In order to remain competitive in the ever-changing world of modern health-care industry, businesses must be flexible and agile. But this flexibility has a cost: there’s a chance that business operations will be disrupted by change. Change management has emerged as a crucial element of organizational success in the fast-paced world of today. To guarantee that changes are executed successfully, efficiently, and with the least amount of disturbance to business operations, a well-structured change management approach is necessary.
A compliance-focused change management solution should: consider the following solutions
- Many organizations have large number of SOPs that cross-relate to changes. Modern organization maintains a centralized queue to manage and confirm incoming change requests from all departments.
- The organization should have well defined SOP, written procedures for identification, ownership, justification for proposed changes. The documentation involved should clearly define, scope and justification of proposed changes; Record all impacted areas, assign tasks, and propose change plans.
- Change management system of the organization should be scalable and has capacity to handle complex changes when necessary.
- Enable one or multiple assessments to allow experts to evaluate the impacts of a change, accounting for results of impact assessments before routing for approval.
- Track and monitor changes in the organization as they are executed and verified.
- Effectiveness of change should be measured holistically at all levels.
- Integrate with other departments regarding new changes to be implemented and provide additional trainings and certifications.
- Incorporate electronic signatures during impact assessment, review, approval within the change process to ensure compliance with 21 CFR Part 11.
Now that you know this: – hopefully you are in position to answer the Assignment(s)
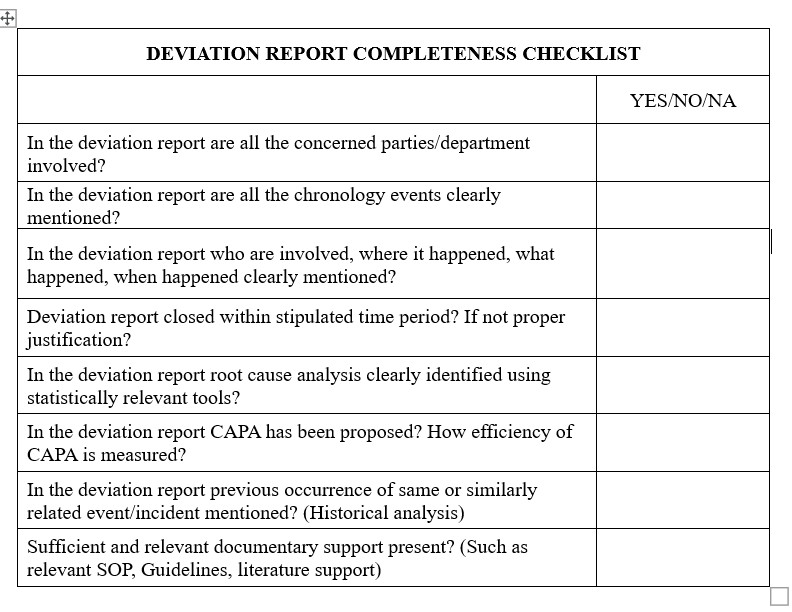
- Do your QA department have SOP for change management, especially related to change control, CAPA, control and uncontrolled deviations, lab incidence and human errors?
- Do your QA/QC department completely use tools such as 5-whys to completely investigate root cause analysis? Do staff have sufficient experience to investigate Root cause analysis?
- Do your QA/QC department sufficiently monitor CAPA efficiency? How they are measured?
- Does your organization have properly assessed proposed changes completely for the risk involved? Classification of changes done?
- Does your organization have centralized SOP control and review system in place? Is it controlled appropriately by change control mechanism to ensure compliance with 21 CFR Part 11?