In today’s rapidly evolving regulatory pharmaceutical landscape, the need for efficient and effective process change controls has become more crucial than ever. As establishments strive to maintain a competitive edge, they must ensure that their change control systems are up-to-date, secure and 21cfr part11 compliance. This short checklist aims to provide a series of thought-provoking questions that will help initiate a robust change control process, ensuring that your organization’s change control systems remain at the forefront in compliance to regulatory guidelines.
1. Is the process new, or a change to an existing one?
2. Has the request been evaluated by the Regulatory department?
3. Has the request been evaluated by the Environmental Health and Safety Department?
4. Has the request been evaluated to establish Validation Recommendations?
5. Requires Characterization (for new Active Pharmaceutical Ingredient [API]):
6. Experimental number necessary?
7. Requires testing of new incoming material?
9. Documentation from the new vendor or Supplier required? Supplier or vendor plant audit necessary? then incorporate Certification of a validated process and Letter of reference and update in Drug Master File.
10. Are there copies of the supplier drawings (current vs. proposed), if components are affected?
11. Is there a comparison of the current and proposed dimensions, if components are affected? Revised component specification?
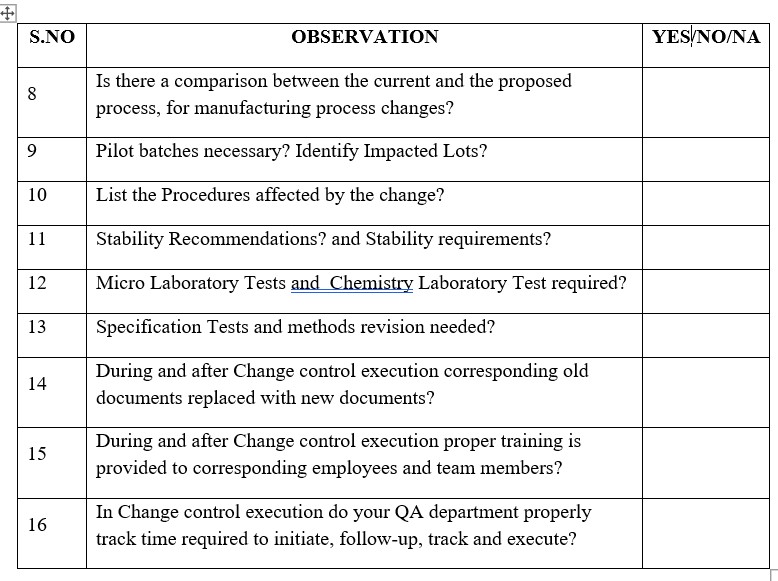
12. Is there a comparison between the current and the proposed process, for manufacturing process changes?
13. Is there a list of finished products impacted, including their respective markets?
14. Pilot batches necessary? and Identify Impacted Lots?
15. Aseptic Process Evaluation needed?
16. Closure Integrity Studies needed?
17. Stability Recommendations?
18. List the Procedures affected by the change?
19. Micro Laboratory Tests/ Chemistry Laboratory Test required?
20. Specification Tests and methods revision needed?
21. Master Batch Record revision needed? (Specify Sections)
22. For Component Changes, is there a Planning Impact Evaluation?
23. Does this Change require an update to Laboratory electronic systems?

After reading this blog, we hope you can understand the importance of change control and properly use the proposed checklist and improve on it further.
Now that you know this:- hopefully you are in position to answer the Assignment(s)
Have you been involved in any change control activities in your lab? If yes recollect your activities?
Does your lab have proper SOP for the implementation of change control in your lab? If so are there effective?
Do you clearly identify whether proposed changed as Critical, Major or Minor? If yes what is the criteria?
Do you clearly identify whether proposed changes effect the quality of the product? Stability of the product? If so then it is properly captured and its impact properly assessed?
What is average time period required to properly close change control in your lab? Can you suggest any 3 measures to improve the quality of change control?
Have you found any communication gaps between different departments to smoothly close change control system? Especially between QA,CRD,ARD and Regulatory departments?